Tosiban 6.75mg & 37.5mg Injection
Therapy Area
Women's Health
Composition

Pharmacotherapeutic Group

Pharmackinetic Properties

Metabolism and Excretion

Clinical Data



Contraindications

Special warnings and precautions for use





Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
Keep this leaflet. You may need to read it again.
If you have any further questions, ask your doctor or pharmacist or nurse.
This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
If you get any side effects, talk to your doctor or pharmacist or nurse. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What TOSIBAN® is and what it is used for
2. What you need to know before you take TOSIBAN®
3. How to take TOSIBAN®
4. Possible side effects
5. How to store TOSIBAN®
6. Contents of the pack and other information
1. What TOSIBAN® is and what it is used for
TOSIBAN® solution for injection contains Atosiban. TOSIBAN® can be used to delay the premature birth of your baby. TOSIBAN® is used in pregnant adult women, from week 24 to week 33 of the pregnancy. TOSIBAN® works by making the contractions in your womb (uterus) less strong. It also makes the contractions happen less often. It does this by blocking the effect of a natural hormone in your body called “oxytocin” which causes your womb (uterus) to contract.
2. What you need to know before you take TOSIBAN®
Do not take TOSIBAN®
- If you are less than 24 weeks pregnant.
- If you are more than 33 weeks pregnant.
- If your waters have broken (premature rupture of your membranes) and you have completed 30 weeks of your pregnancy or more. If your unborn baby (foetus) has an abnormal heart rate.
- If you have bleeding from your vagina and your doctor wants your unborn baby to be delivered straight away.
- If you have something called “severe pre-eclampsia” and your doctor wants your unborn baby to be delivered straight away. Severe pre-eclampsia is when you have very high blood pressure, fluid retention and/or protein in your urine.
- If you have something called “eclampsia” which is similar to “severe pre-eclampsia” but you would also have fits (convulsions). This will mean your unborn baby needs to be delivered straight away.
- If your unborn baby has died.
- If you have or could have an infection of your womb (uterus).
- If your placenta is covering the birth canal.
- If your placenta is detaching from the wall of your womb.
- If you or your unborn baby have any other conditions where it would be dangerous to continue with your pregnancy.
- If you are allergic to TOSIBAN® or any of the other ingredients.
Do not use TOSIBAN® if any of the above apply to you. If you are not sure, talk to your doctor, midwife or pharmacist before you are given TOSIBAN®.
Warnings and precautions
Talk to your doctor or pharmacist or nurse before using TOSIBAN®:
If you think your waters might have broken (premature rupture of your membranes).
If you have kidney or liver problems.
If you are between 24 and 27 weeks pregnant.
If you are pregnant with more than one baby.
If your contractions start again, treatment with TOSIBAN® can be repeated up to three more times.
If your unborn baby is small for the time of your pregnancy. Your womb may be less able to contract after your baby has been born. This may cause bleeding.
If you are pregnant with more than one baby and/or are given medicines that can delay the birth of your baby, such as medicines used for high blood pressure. This may increase the risk of lung oedema (accumulation of fluid in the lungs).
If any of the above apply to you (or you are not sure), talk to your doctor, midwife or pharmacist before you are given TOSIBAN®.
Children and adolescents
TOSIBAN® has not been studied in pregnant women less than 18 years old.
Other medicines and TOSIBAN®
Tell your doctor or pharmacist if you are using, have recently used or might use any other medicines.
Pregnancy, breast-feeding and fertility
You should only be given this medicine if you are between 24 and 33 weeks of pregnancy. If this does not apply to you, please speak to your doctor. If you are still breast feeding a child from an earlier pregnancy, you should stop during your treatment with TOSIBAN®, as breastfeeding may stimulate uterine contractions.
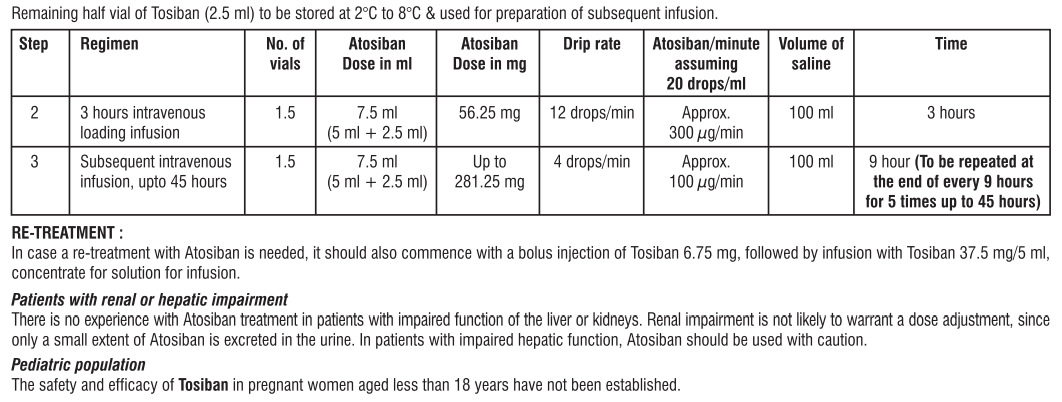
3. How to use TOSIBAN®
TOSIBAN® will be given to you in a hospital by a doctor, nurse or midwife. They will decide how much you need. They will also make sure the solution is clear and free from particles.
TOSIBAN® will be given into a vein (intravenously) in three stages:
- The first injection of 6.75 mg in 0.9 ml will be slowly injected into your vein over one minute.
- Then a continuous infusion (drip) will be given at a dose of 18 mg per hour for 3 hours.
- Then another continuous infusion (drip) at a dose of 6 mg per hour will be given for up to 45 hours, or until your contractions have stopped.
Treatment should last no longer than 48 hours in total. Further treatment with TOSIBAN® can be used if your contractions start again. Treatment with TOSIBAN® can be repeated up to three more times. During treatment with TOSIBAN®, your contractions and your unborn baby’s heart rate may be monitored. It is recommended that no more than three re-treatments should be used during a pregnancy.
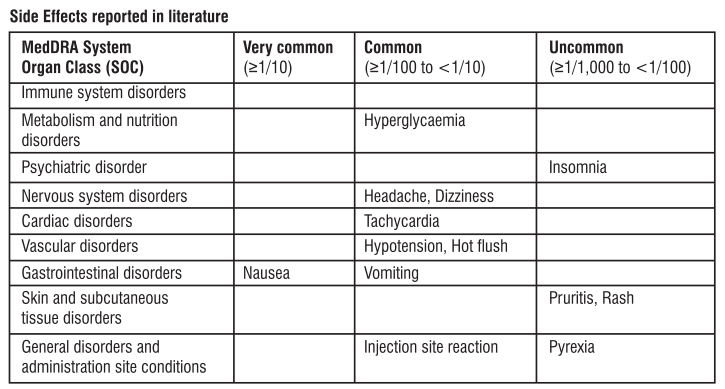
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. The side effects seen in the mother are generally of a mild severity. There are no known side effects on the unborn or new-born baby.
The following side effects may happen with this medicine:
Very common (affects more than 1 in 10 people)
- Feeling sick (nausea).
Common (affects less than 1 in 10 people)
- Headache.
- Feeling dizzy.
- Hot flushes.
- Being sick (vomiting).
- Fast heart beat.
- Low blood pressure.Signs may include feeling dizzy or light-headed.
- A reaction at the site where the injection was given.
- High blood sugar.
Uncommon (affects less than 1 in 100 people)
- High temperature (fever).
- Difficulty sleeping (insomnia).
- Itching.
- Rash.
Rare (affects less than 1 in 1,000 people)
- Your womb may be less able to contract after your baby has been born. This may cause bleeding.
- Allergic reactions.
You may experience shortness of breath or lung oedema (accumulation of fluid in the lungs), particularly if you are pregnant with more than one baby and/or are given medicines that can delay the birth of your baby, such as medicines used for high blood pressure.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly: Website: www.zuventus.co.in and click the tab “Safety Reporting” located on the top right end of the home page. By reporting side effects, you can help provide more information on the safety of this medicine. You can also report the side effect with the help of your treating physician.
5. How to store TOSIBAN®
Keep this medicine out of the sight and reach of children. Do not use TOSIBAN® after the expiry date (EXP:) which is stated on the pack. The expiry date refers to the last day of that month. Store in a refrigerator (2°C - 8°C). Store in the original package in order to protect from light. Once the ampoule has been opened, the product must be used straight away. Do not use this medicine if you notice particulate matter and discoloration prior to administration.
6. Contents of the pack and other information
What TOSIBAN® contains
Tosiban 6.75 mg Solution for Injection
Each 0.9 ml contains
Atosiban Acetate IP equivalent to Atosiban 6.75 mg
Water for Injections IP q.s.
Tosiban 37.5 mg Concentrate for Solution for Infusion
Each 5 ml contains
Atosiban Acetate IP equivalent to Atosiban 37.5 mg
Water for Injections IP q.s.